Cytokeratin 5 Antibody – 165Ho
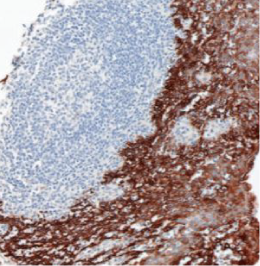
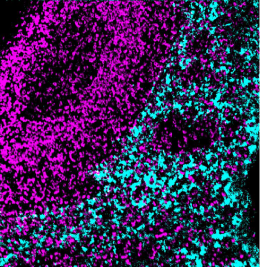


Validation: Each lot of conjugated antibody is quality control tested by staining tissue following the MIBI Staining Protocol optimized for the applicable tissue format with subsequent MIBIscope analysis using the appropriate positive and negative tissue field of views. These results are pathologist verified.
Recommended Usage: Human FFPE: 1:100 dilution.
For optimal results, the antibody should be titrated for each desired application.
MIBI technology: Learn more about MIBI™ Technology, a multiplex IHC technology with unmatched sensitivity and true subcellular resolution.
References
Silvia Mihaela Illie et al. Potential clinically useful prognostic biomarkers in triple-negative breast cancer: preliminary results of a retrospective analysis. Breast Cancer (Dove Med Press). 2018; 10: 177–194.
Corr BR et al. Cytokeratin 5-positive cells represent a therapy resistant subpopulation in epithelial ovarian cancer. Int J Gynecol Cancer. 2015 Nov;25(9):1565-73.
* Conjugate tested on human tissue.